Mechanism of Action for WELIREG® (belzutifan)
WELIREG is an inhibitor of hypoxia-inducible factor 2 alpha (HIF-2α)
Under normal oxygen levels, HIF-2α is targeted for degradation by VHL protein
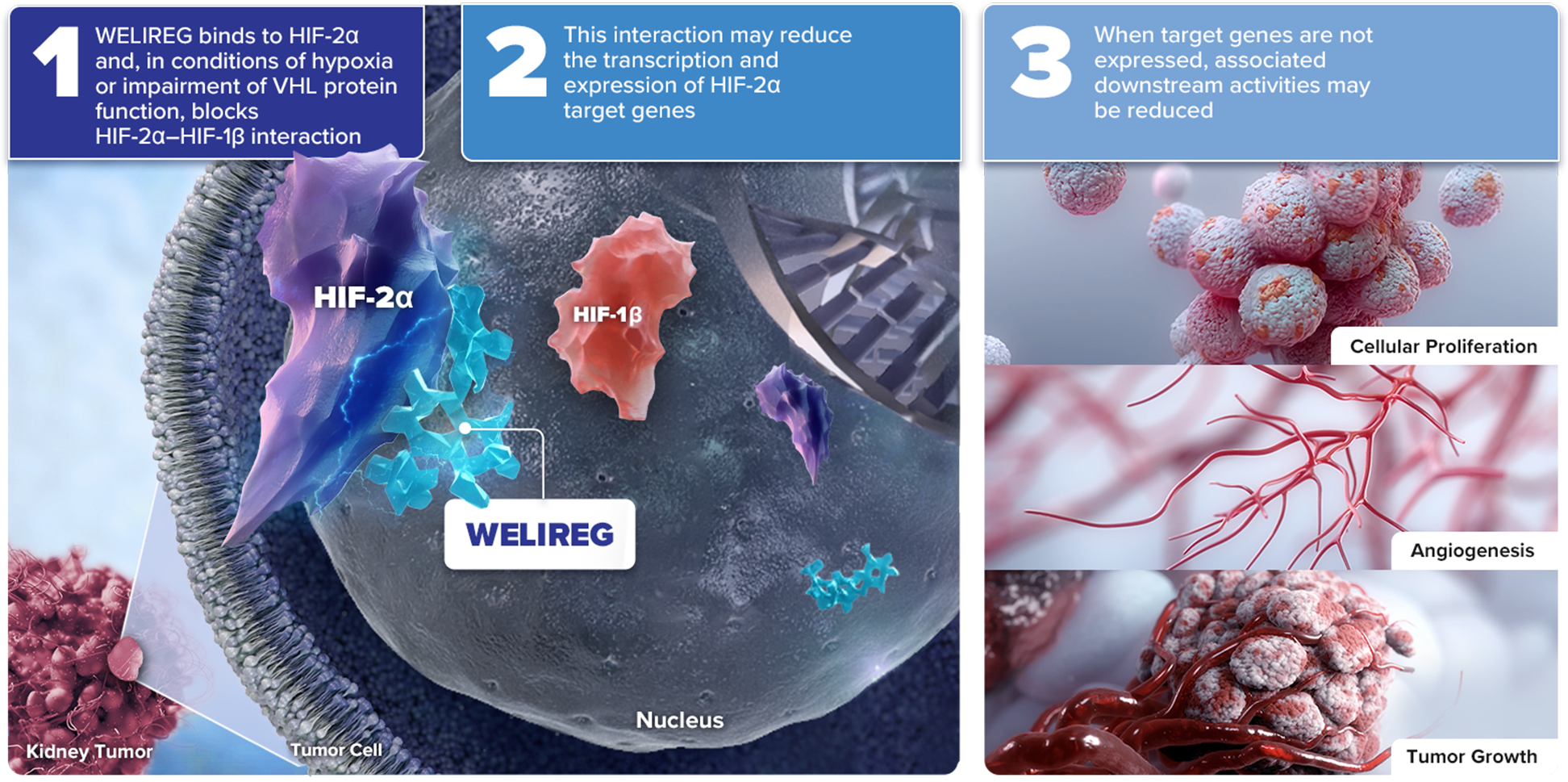

As a selective HIF-2α inhibitor, WELIREG is the only approved systemic management option for adult patients with certain VHL disease–associated tumors, when surgery is not immediately required1,2
References: 1. Narayan V, Jonasch E. Systemic therapy development in von Hippel-Lindau disease: an outsized contribution from an orphan disease. Cancers (Basel). 2022;14(21):5313. 2. Center for Drug Evaluation and Research. Advancing Health Through Innovation: New Drug Therapy Approvals 2021. https://www.fda.gov/media/155227/download. Accessed October 15, 2025.